Effective April 1, 2024, healthcare entities operating in California face a new set of requirements from state regulators. The Health Care Quality and Affordability Act (HCQAA) requires healthcare entities to provide written notice of an agreement or transaction to the newly created Office of Health Care Affordability (OHCA), which will evaluate the transaction’s costs and market impacts to determine whether the transaction threatens market competition or healthcare affordability. In addition, expenditures by healthcare entities will be monitored, and cannot exceed the statewide cost targets established by OHCA on an annual basis.
Key issues for companies operating in the healthcare space:
Which entities are required to provide notice to OHCA?
“Healthcare entities” operating in California, which include: payors; fully integrated delivery systems; physician organizations (specifically, those with 25 or more physicians or that otherwise are considered “high-cost outliers”); risk-bearing organizations; health facilities (such as general acute care hospitals, acute psychiatric hospitals, nursing facilities, intermediate long-term care facilities, and hospice); 1206(d) hospital outpatient departments; 1206(l) medical foundations; 1204(a) community clinics; 1204(b) specialty clinics; ambulatory surgery centers; clinical laboratories; and imaging facilities.
Which entities are NOT required to provide notice to OHCA?
Physician organizations with fewer than 25 physicians that are not deemed “high-cost outliers”; management services organizations (MSOs) and other business support entities that provide administrative or other services to providers; non-physician healing arts organizations (e.g., dental corporations and chiropractic corporations); pharmacies; and providers that are not specified under the law, such as 1206(g) teaching clinics.
What transactions are implicated?
Agreements and transactions that transfer a “material amount” of a healthcare entity’s assets or operational control. Notably, to the extent that the agreement or transaction does not include direct investment in, or acquisition of, a healthcare entity, these transactions are currently exempt from the notice requirement. In addition, transactions already subject to other California regulatory review processes (e.g., nonprofit health facility transactions that require approval from the California Attorney General or health plan transactions subject to the oversight of the California Department of Managed Health Care) are generally exempt.
What factors are evaluated by OHCA for implicated transactions?
Although HCQAA does not identify the specific factors and standards considered by OHCA during its evaluation of implicated transactions for the creation of its preliminary and final reports, the statute identifies that OHCA will adopt regulations addressing its review process and relevant standards.
What conditions may OHCA impose on these transactions?
HCQAA does not require that OHCA provide explicit approval for a transaction; however, approval is implicit within the notice process. And, while the statute does not specify how OHCA will treat transactions that it finds objectionable based on its final cost and market impact report (i.e., whether these transactions will be blocked or if conditions may be imposed), future regulations may further clarify OHCA’s scope of authority.
How else is OHCA monitoring and enforcing healthcare expenditures other than on a transaction-specific basis?
On an annual basis starting in 2025, OHCA will establish a statewide healthcare cost target. Spending by healthcare entities cannot be greater than OHCA’s annual cost target. Healthcare entities must submit annual reports with data and information to OHCA to allow OHCA to set annual targets and monitor compliance.
When do these requirements take effect, and how does this impact deal timelines?
- As of April 1, 2024, healthcare entities must provide notice of applicable transactions.
- As of September 1, 2024, certain healthcare entities must start reporting data to OHCA.
- Commencing in 2026, OHCA may enforce compliance with OHCA’s healthcare cost targets.
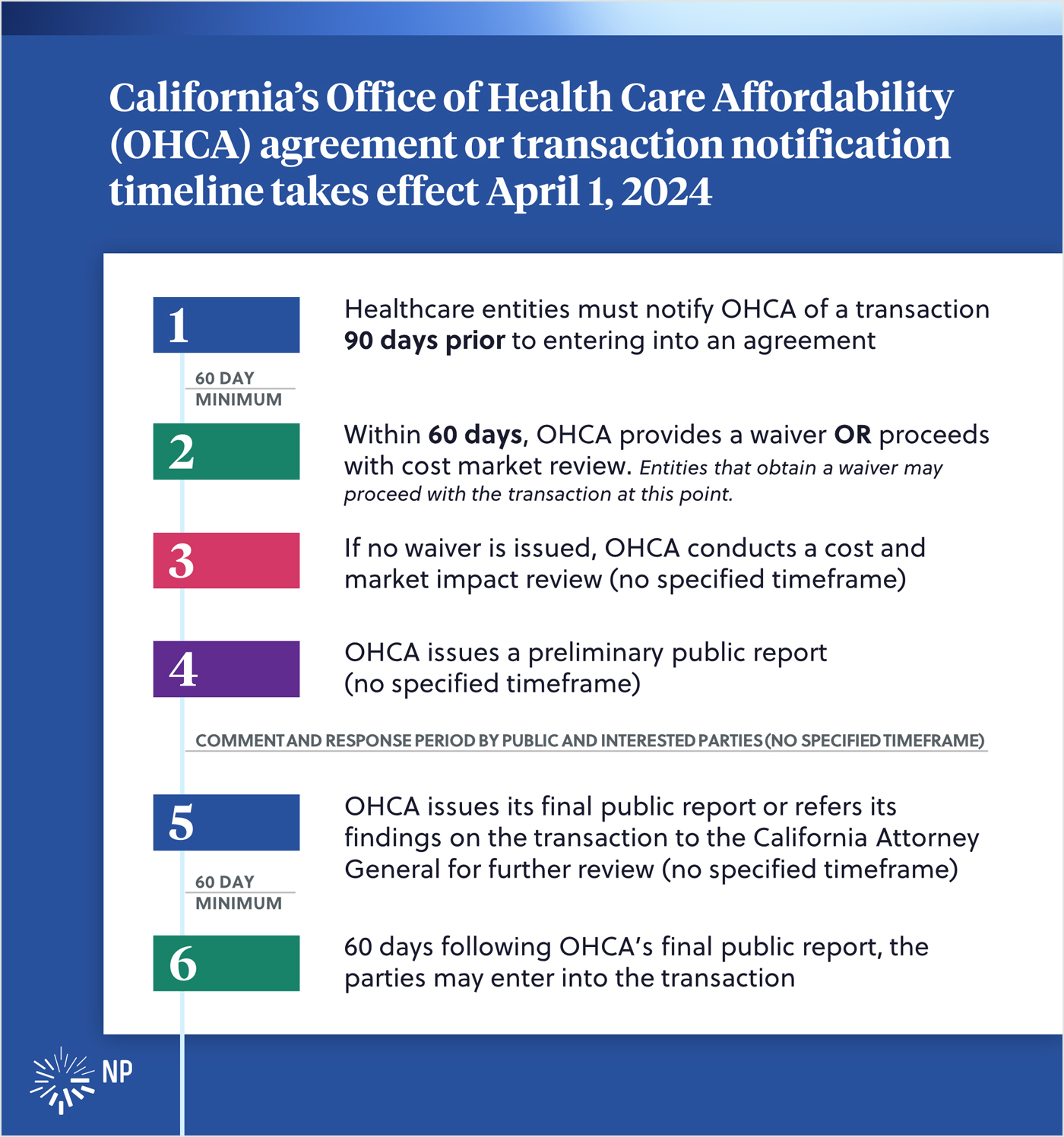
- The initial OHCA review process will require at minimum, a 60-day review period. If OHCA does not provide a waiver of the transaction, the review process will be extended—the current statute does not specify the total timeframe allotted for this extension, and without further regulatory guidance, at minimum, includes an additional 60-day waiting period.
We expect to see a flurry of deal activity over the next year as healthcare entities that will be impacted by these requirements race to close transactions or contemplate restructuring in advance of HCQAA’s April 1, 2024, effective date. Following that, we anticipate that healthcare entities and their affiliates will seek to structure their operations, transactions, and affiliations in a manner that limits regulatory filings and reporting.
Additionally, although healthcare companies have been largely focused on the impact of the transaction-specific reporting requirements, it is critical to note that all healthcare entities (as such term is defined under HCQAA) will also be subject to annual reporting requirements and will be monitored to ensure compliance with OHCA’s annual statewide “healthcare cost target,” which is intended to limit healthcare spending to an established targeted cost of expenditures. Exceeding these cost targets may result in corrective actions and the potential for administrative fines.
Based on the OHCA Board’s 2023-2024 12-Month Workplan, we anticipate regulations will be issued by September 2023 that will provide further guidance and understanding of the potential implications of HCQAA.
Below, we have provided additional details about how these requirements impact healthcare entities.
Requirements for notice by healthcare entities of certain transactions
“Healthcare entities” defined
HCQAA’s notice requirement applies to “healthcare entities,” which is defined as a payor, provider, or a fully integrated delivery system.[1] The definition of “provider” includes the following: physician organizations of 25 or more physicians; physician organizations with fewer than 25 physicians that are deemed “high-cost outliers” (meaning, services provided by that physician organization are substantially higher compared to the statewide average, as assessed by OHCA); risk-bearing organizations; health facilities (which include general acute care hospitals, acute psychiatric hospitals, nursing facilities, intermediate long-term care facilities, and hospice); 1206(d) hospital outpatient departments; 1206(l) medical foundations; 1204(a) community clinics; 1204(b) specialty clinics; ambulatory surgery centers; clinical laboratories; and imaging facilities.
In light of the HCQAA’s definition of “healthcare entities,” OHCA’s notice requirements do not appear to apply to the following entities: physician organizations with fewer than 25 physicians that are not “high-cost outliers”; MSOs and other business support entities that provide administrative or other services to providers; non-physician healing arts organizations (e.g., dental corporations and chiropractic corporations); pharmacies; and providers that are not specified under the law, such as 1206(g) teaching clinics.
Thus, absent further guidance under the forthcoming regulations, transactions that are structured as an investment in, or acquisition of, commercial business entities that merely operate in the healthcare arena do not appear to trigger OHCA’s notice requirements because these companies do not qualify as “healthcare entities.” For example, investment in an MSO that manages physician organizations or other healthcare entities (such as those operating a healthcare enterprise through a PC-MSO model) appears to be exempt from the notice requirements because an MSO does not meet the definition of “healthcare entity.” In addition, organizations that provide services in non-physician healing arts specialties do not qualify as “healthcare entities,” and thus, are exempt from the requirements. Further, entities that are already subject to other California regulatory review processes are generally exempt from OHCA notification requirements (unless specifically referred to OHCA for review), including Knox-Keene health plans regulated by the California Department of Managed Care, and nonprofit corporations already subject to review by the California Attorney General (currently facing a legislative challenge via SB 774).
Transactions that trigger notification
Notice to OHCA must be provided if a healthcare entity enters into an agreement or transaction that involves either of the following:
- sale, transfer, lease, exchange, option, encumbrance, conveyance, or disposal of a material amount of a healthcare entity’s asset to one or more entities; or
- transfer of control, responsibility, or governance of a material amount of the assets or operations of the healthcare entity to one or more other entities.
Notably, the HCQAA does not define what constitutes a “material amount,” but the regulations may provide further definition around this threshold. At this time, our expectation is that a material amount would include customary indicators of materiality, such as a sale of substantially all of the assets of a healthcare entity, and a transfer of majority rights over governance control or profits.
Content of notification; confidentiality of sensitive information
The HCQAA does not specify the requirements for the content of the notification provided to OHCA. As a result, the law’s implementing regulations will be critical in understanding the scope of the materials that will be produced regarding the healthcare entity and proposed transaction.
Under the HCQAA, OHCA is required to keep all nonpublic information obtained during the review process confidential. However, OHCA may disclose nonpublic information in its preliminary and final reports that it believes to be in the public interest, subject to considerations about privacy, trade secret, and anticompetitive factors. Because notification is required in advance of the parties’ entrance into an agreement, parties will likely be required to disclose information about the proposed transaction at early stages of negotiation. Such disclosures may stifle the appetite for healthcare entities to engage in transactions that may expose sensitive or propriety business information to competitors and affiliates—especially if there is a concern that this will undermine current and future strategic planning. Undoubtedly, this will impact how parties approach deal negotiations and transaction timelines.
Timelines: Cost and market review process
OHCA’s cost and market review process includes key timeframes, described below. Importantly, there is still some uncertainty around the total timeline associated with the review process, which should be considered when projecting deal timeframes:
- At least 90 days prior to “entering into an agreement,” healthcare entities must notify OHCA of the transaction. The statute does not further define the meaning of “entering into an agreement.”
- Within 60 days of receipt of the notice, OHCA determines whether it will conduct a cost and market impact review or issue a waiver from the cost and market review. If provided a waiver from cost and market review, the transaction may proceed.
- If OHCA determines the transaction or other material change is likely to have a significant impact on market competition, the state’s ability to meet cost targets, or costs for purchasers and consumers, OHCA conducts a cost and market impact review. The review considers, among several factors, whether the transaction would result in increased access to services, higher quality services, and more efficient services to consumers. Neither the HCQAA nor any released guidance provides a timeframe for OHCA’s cost and market review nor the methodology for establishing standards for OHCA’s review.
- Upon completion of the review, OHCA issues a preliminary public report.
- Affected parties and the public have an opportunity to respond to the preliminary public report. No timeframe is specified for the response period.
- OHCA issues its final cost and market review public report. OHCA may also refer its findings on the transaction to the California Attorney General for further review of any unfair methods of competition, anticompetitive behavior, or anticompetitive effects. No timeframe is specified for the completion of the final public report, and the statute does not specify whether OHCA will impose conditions on the transaction as part of the final report.
- 60 days following the issuance of OHCA’s final public report, the parties may enter into the transaction.
Data reporting; statewide cost targets
The HCQAA also creates new reporting requirements for healthcare entities. By September 1, 2024, payors and fully integrated delivery systems are required to submit data on total healthcare expenditures covering calendar years 2022 and 2023. In addition, the HCQAA requires other healthcare entities to submit data and other information deemed necessary by OHCA to measure total healthcare expenditures, per capita total healthcare expenditures, standard quality measures, alternative payment models, primary care, and behavioral health expenditures (although a specific date for these reporting requirements is not identified under the law, we anticipate this to be prior to 2025).
Importantly, the data reported by healthcare entities will be utilized by OHCA to set statewide healthcare cost targets and baselines on an annual basis starting in 2025 and each subsequent calendar year. These healthcare cost targets will be based on healthcare sectors, which may take into account geographic regions and type of healthcare entity, and will represent the target percentage for the maximum annual increase in per capita total healthcare expenditures.
In other words, the state will identify and enforce what is in effect a cap on total annual healthcare expenditures by each healthcare entity.
Although 2025 serves as the first reporting year for healthcare entities, the HCQAA states that OHCA will not enforce compliance with cost targets until 2026. If a healthcare entity exceeds the applicable healthcare cost target, OHCA will notify the entity and provide it with a 45-day response period to identify the reasons for exceeding the cost target. If OHCA determines that the entity is not compliant and not otherwise excused from compliance, it may take certain actions to provide technical assistance, require public testimony by entities regarding the failure to meet cost targets, facilitate performance improvement plans, and levy administrative penalties.
Other states
Other states have taken similar action to bolster state oversight over the provision of clinical care, including over transactions and commercial investment in the healthcare space. In addition to California, a number of states—Connecticut, Delaware, Massachusetts, Nevada, New Jersey, New York, Oregon, Rhode Island, and Washington—have established new, independent commissions or have otherwise expanded the authority of an existing regulatory body to monitor and limit growth in health spending. These laws limit or restrict M&A activity with a goal of addressing healthcare competition and accessibility issues, tying the analysis to transactional activity.
Looking ahead
According to the HCQAA and FAQs released by OHCA, OHCA has indicated that it will publish regulations in 2023 that outline key provisions of the HCQAA, including the material changes that warrant notification, thresholds for determining which healthcare entities or prospective transactions will be subject to the notice requirements, factors to be considered in market and cost reviews, requests for data, and relevant timelines.
Our Healthcare team will be closely tracking the release of the accompanying regulations as additional information is needed to understand the full scope and impact that OHCA will have on the California healthcare landscape. We continue to monitor similar legislation in other states, and how these actions will affect entities operating in the healthcare arena across state lines.
- “Fully integrated delivery system” means a system that includes a physician organization, health facility or health system, and a nonprofit healthcare service plan that provides healthcare services to enrollees in a specific geographic region through an affiliate hospital system and an exclusive contract between the nonprofit healthcare service plan and a single physician organization in each geographic region to provide those medical services.
[back to reference ]
